Healing the Ozone Layer Through Diplomacy
Still Only One Earth: Lessons from 50 years of UN sustainable development policy
The Montreal Protocol shows what is possible when science, diplomacy, and business cooperate to implement international environmental agreements. When the people became aware of a hole in the ozone layer over Antarctica—and damage to the protective layer of atmosphere around the world—leaders around the world coordinated action on ozone depleting substances. (Download PDF) (See all policy briefs) (Subscribe to ENB)
When scientists discovered a massive hole in the ozone over Antarctica in 1985, the world was struck with fear. Public health experts immediately warned the increasing intensity of UV radiation that now penetrates the atmosphere may greatly increase the incidence of skin cancer and cataracts, and could significantly damage global crops and the marine food chain. Ozone forms a protective layer over the Earth, absorbing dangerous UV radiation, which can harm each and every one of us.
As news of the ozone hole spread through the media, it became a worldwide sensation. Scientists scrambled to understand the chemical processes behind the hole as the public expressed fear for the scientists working at the South Pole, assuming they would be exposed to UV rays that could render them blind and sunburned. Rumours of blind sheep spread due to concerns the increased radiation would cause cataracts—and potential for increased skin cancer also stoked public fears (Blakemore, 2016).
But through the Vienna Convention for the Protection of the Ozone Layer and the Montreal Protocol on Substances that Deplete the Ozone Layer, great steps have been taken to heal the ozone layer. But is it enough?
What is the Ozone Layer and Why is it Important?
Ozone depletion was not on the agenda at the Stockholm Conference on the Human Environment in 1972. Yet shortly thereafter, scientists discovered certain human-made chemicals posed a serious threat to the Earth’s ozone layer.
Ozone is a pungent, slightly bluish gas composed of three oxygen atoms (O3). Nearly 90% of naturally occurring ozone resides in the stratosphere, the portion of the atmosphere ten to fifty kilometres (six to thirty miles) above the Earth. Commonly called the ozone layer, stratospheric ozone helps to shield the planet from UV radiation. Even though only about three of every ten million molecules in the atmosphere are ozone, the ozone layer absorbs all the deadly UV-C radiation and most of the harmful UV-B radiation emitted by the sun. UV-B and UV-C denote electromagnetic radiation of different wavelengths (Chasek & Downie, 2021).
Scientists Mario Molina and F. Sherwood Rowland published a paper in 1974 showcasing their Nobel-prize winning research that chlorofluorocarbons (CFCs) cause ozone depletion. Their research showed CFCs release chlorine atoms into the stratosphere that act as a catalyst in the destruction of ozone molecules. In fact, one chlorine molecule can destroy over 100,000 ozone molecules before it is removed from the stratosphere (US EPA, n.d.). Furthermore, they found CFCs can live for decades in the atmosphere. Created in the 1920s to replace flammable and noxious refrigerants, CFCs are inert, nonflammable, nontoxic, colorless, odorless, and adaptable to a wide variety of uses. By the mid-1970s, CFCs had become the chemical of choice for coolants in refrigeration and air-conditioning, propellants in aerosol sprays, solvents in the cleaning of electronic components, and the blowing agent for the manufacture of flexible and rigid foam (Chasek & Downie, 2021). The popular gases were not as innocent as they seemed, however, and further research has shown this extends to include compounds containing bromine, such as methyl bromide and halons (Salawitch et al., 2019), as well as other chemicals like carbon tetrachloride and methyl chloroform.
The years following the publication of our paper were hectic, as we had decided to communicate the CFC–ozone issue not only to other scientists, but also to policy makers and to the news media; we realized this was the only way to ensure that society would take some measures to alleviate the problem.
It took coordinated international action to avoid a scenario where the world would have suffered both negative ozone and climate effects. The adoption of the 1985 Vienna Convention for the Protection of the Ozone Layer and the 1987 Montreal Protocol on Substances that Deplete the Ozone Layer helped to avoid two million cases of skin cancer per annum or, by 2100, an additional 443 million cases of skin cancers and 2.3 million skin cancer deaths in the United States alone, including 8-10 million cases of malignant melanoma. These treaties are also credited with the avoidance of 63 million additional cataract cases, plus a 6% global reduction in plant production for each 10% loss in ozone (Fahey, 2013). The Montreal Protocol also delayed the increase in climate forcing—the change in globally averaged temperature changes due to natural or human-induced activities—by 7-12 years (Molina et al., 2009; CAMS, n.d.).
The Role of the Vienna Convention and the Montreal Protocol
The story of the ozone layer is one of multilateralism’s great successes. Molina and Roland’s research showed CFCs can break down the ozone layer, and along with the confirmation of an ozone hole above the Antarctic by British scientists Joseph Farman, Brian Gardiner and Jonathan Shanklin in 1985 (Birmpili, 2018), were the catalysts that spurred the international community to coordinate an effective response to the shared crisis.
The first step was the World Plan of Action on the Ozone Layer, adopted by the United Nations Environment Programme (UNEP) in 1977. This plan, which called for intensive international research and monitoring of the ozone layer, was followed in 1981 by a decision to draft a global framework convention on stratospheric ozone protection (Weiss, 2009). This ultimately led to the adoption of the Vienna Convention for the Protection of the Ozone Layer in 1985.
The Vienna Convention was a dramatic step forward to protect the ozone layer. However, many were disappointed it did not feature controls but rather focused on research, cooperation, and monitoring (Hajost & Koehler, 1990). Spurred on by the discovery of the Antarctic ozone hole, just two years later in 1987, the Montreal Protocol on Substances that Deplete the Ozone Layer did make the leap to include control measures for CFCs and halons.
The Montreal Protocol has had great success, particularly as it is one of the first international agreements that addressed a problem caused by actions in the present, but where the effects would only be seen years, if not decades, later (Weiss, 2009). It operates effectively at the nexus of science, diplomacy, and the private sector. It has also been cleverly crafted so should scientific evidence show further action is needed, adjustments and amendments can be made. This option has been used to great effect with the London, Copenhagen, Montreal, Beijing and, most recently, the Kigali Amendments.
The Protocol’s decisions have always been taken based on sound science. The Protocol has three assessment panels—the Technology and Economic Assessment Panel, the Scientific Assessment Panel, and the Environmental Effects Assessment Panel—which provide the necessary expertise to investigate and review the latest scientific developments.
When negotiating the Protocol, countries also recognized two classes of parties—Article 2 and Article 5 parties. The former are those parties with the monetary and technical capacity to effect changes in consumption and production of ozone depleting substances (ODS). The latter are those parties who need assistance in meeting their obligations and lack the necessary economic and technical resources and are generally considered to be “developing countries.” Through recognizing these “common but differentiated responsibilities,” and by establishing the Multilateral Fund to help provide financial resources to address these insufficiencies, the Protocol wanted to ensure all countries were able to comply with their obligations to protect the ozone layer. Parties also set differentiated phase-out and phase-down schedules for production and consumption of ODS. Through these two interventions, the differentiated circumstances of all parties are recognized and addressed, contributing to a more even playing field. These steps have also helped ensure equal commitment from all parties as they strive to phase out ODS.
The private sector has also been key to ensuring success. Following the discovery of the damage ODS can wreak on the ozone layer, and the publication of the first comprehensive scientific assessment of the issue, DuPont, the leading CFC manufacturer at the time, called for the complete phase-out of CFCs and committed themselves to doing so, closing their last production facility in 1999 (McFarland, 2009; Dupont, 2019). As more amendments have been added to the Protocol, more companies have come on board to assist in meeting the Protocol’s obligations. For example, during the late 1980s, the National Fire Protection Association, on behalf of the industry, undertook research to find low-ODS alternatives to halons. More recently, Daikin has granted free access to patents for technology using low ozone-depleting potential substances. In the run-up to the adoption of the Kigali Amendment, which addresses hydrofluorocarbons (HFCs), Coca-Cola announced its goal to ensure new coolers and vending machines would be HFC-free by 2015.
Lastly, political will has been central to ensuring international agreement for necessary action. Following the discovery of the ozone hole, many countries took swift national action. For example, the US banned non-essential use of CFCs in aerosols; Sweden, Norway, and Denmark prohibited the use of CFCs in propellants; and the Netherlands mandated warning labels on all products (Ott, 1991). As Ott (1991) notes, pressure from the US drove governments such as Japan and the European Commission to reach an agreement on the Protocol. Amendments to include further controls, in particular the Kigali Amendment to address HFCs, and the establishment of the Multilateral Fund only serve to underscore the political will that has helped lead this success story. The wide ratification of the Vienna Convention and the Montreal Protocol by 198 countries testifies to the potential for a few influential states to act as catalysts to action.
Chemicals Controlled by the Montreal Protocol
| Chemical | Use | Controls |
|---|---|---|
| Chlorofluorocarbons (CFCs) | refrigeration and air conditioning systems; in spray cans and metred-dose inhalers as aerosol propellants; to make flexible and rigid foams (e.g. seat cushions and Styrofoam); in solvents | 1987 Montreal Protocol and 1990 London Amendment |
| Halons | fire extinguishing systems | 1987 Montreal Protocol |
| Carbon Tetrachloride | solvent or cleaning agent but also in fire extinguishers and as an industrial chemical, including in the creation of refrigerants | 1990 London Amendment |
| Methyl chloroform | solvent | 1992 Copenhagen Amendment |
| Hydrochlorofluorocarbons (HCFCs) |
Air conditioning systems; foam blowing and solvents | 1992 Copenhagen Amendment |
| Methyl bromide | Pesticide and insecticide | 1992 Copenhagen Amendment |
| Hydrobromofluorocarbons | Refrigerant | 1999 Beijing Amendment |
| Hydrofluorocarbons (HFCs) | Refrigeration and air conditioning systems; fire extinguishing systems; insulation; aerosols | 2016 Kigali Amendment |
Climate and Ozone Hand-in-Hand
From the outset, the ozone regime has played a large role in combatting both climate and ozone issues. While CFCs have been best known for their ozone-depleting potential, they also have significant global warming potential and have contributed to climate change. The Protocol has therefore had significant climate co-benefits; by phasing out these substances, greenhouse gas emissions have also decreased. In 2010, emissions reductions due to the Montreal Protocol ranged from 9.7 to 12.5 gigatonnes of CO2 equivalent—approximately five to six times larger than the Kyoto Protocol target (Fahey, 2013; McFarland, 2009).
These climate co-benefits have only been enhanced through the adoption and entry into force of the Kigali Amendment. HFCs are used primarily in air-conditioning systems, where they replaced HCFCs, which had been added to the Protocol’s control mechanisms under the 1992 Copenhagen Amendment. While not ODS, HFCs have a high global warming potential (GWP) and resulted from the phase-out of HCFCs as part of the implementation of the Montreal Protocol and its amendments. HFCs have low ozone-depleting potential and thus seemed to make a good substitute for HCFCs. Parties did not take their impact on the climate into account.
Governments are now set out on a path that will decrease the use of substances that do not normally fall under the remit of the Protocol, save for the fact the HFC “problem” was caused by the phase-out of other ODS. Indeed, the adoption of the Kigali Amendment potentially offers one big climate impact: the world would avoid 0.4°C of global warming by 2100 (Ozone Secretariat, 2020).
The Montreal Protocol stands out as proof positive that the Earth’s nearly 200 countries can effectively cooperate to protect their citizens from a planetary pollution crisis—address climate change as well as ozone depletion. We saved the ozone layer. We can save the climate.
Many see these co-benefits as underscoring the success of the Protocol, and the ozone regime as a whole. The ongoing and highly politicized nature of the climate negotiations have sought some to seek refuge and strive for climate gains under the ozone regime. What has been clear from the outset, and underscored throughout the negotiations of the Kigali Amendment, is the ozone regime serves to address ozone depletion and its consequences. Negotiators assured concerned parties that while climate benefits are welcome, they are simply that—benefits. They are not the primary focus of the regime.

Continuing to Ensure Life on Earth
By successfully implementing the Montreal Protocol, governments have taken concrete steps to protect life on Earth and, in some ways, “reset” the ozone layer. The control measures under the Protocol will in fact return the ozone layer to pre-1980 levels. According to the World Meteorological Organization (2019), the ozone layer over most of the northern hemisphere and mid-latitudes should fully recover by the 2030s, over the southern hemisphere in the 2050s, and above Antarctica later this century (WMO, 2019). This, along with the boon of climate co-benefits, can help address some of the myriad of problems currently facing the planet.
Despite the success of the Montreal Protocol thus far, there is still more work to be done. Although most ODS are being phased out, the Montreal Protocol still allows some quantities of ODS to be produced and used for certain applications through essential use exemptions. Cost-effective alternatives to these applications need to be developed. The use of HFCs also continues, even though environmentally friendly alternatives exist. The Montreal Protocol needs to adopt additional measures to further limit the use and emissions of HFCs. Finally, large quantities of ODS can still be found in old equipment such as refrigeration and air conditioning systems. These chemicals will be released into the atmosphere if not properly reclaimed and destroyed. Measures enabling the safe recovery of remaining ODS are essential (European Environment Agency, 2020).
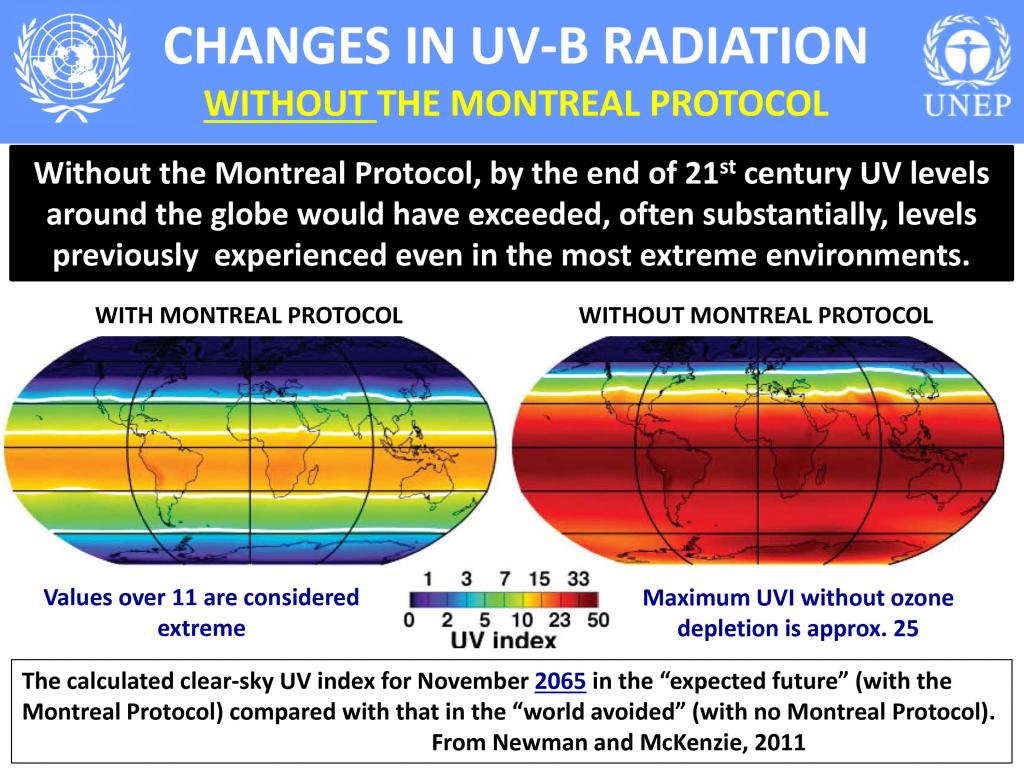
The ozone regime has been instrumental in ensuring life on Earth remains largely unharmed. This has occurred despite the intangibility of the issue—ozone depletion and its future effects are not as obvious as some other environmental issues, such as overfishing or marine pollution. Even with this challenge, the science has been well communicated and incontrovertible: continued uncontrolled ODS use will cause untold harm to the health of the planet and all its living creatures, including humans.
The Montreal Protocol provides a sound example of an international environmental agreement that works. Successfully operating at the nexus of science, diplomacy, and the private sector for over three decades demonstrates that the model is effective, especially when replacements for ODS are available. This also provides the hope that other urgent environmental issues can be resolved swiftly and amicably, particularly if all stakeholders work together.
As Mario Molina, in his Nobel lecture, stated “[ozone depletion] has…shown us that different sectors of society can work together—the scientific community, industry, environmental organizations, government representatives and policy makers—to reach international agreements: the Montreal Protocol on Substances that Deplete the Ozone Layer has established a very important precedent for the solution of global environmental problems” (Molina, 1995).

Works Consulted
Birmpili, T. (2018). Montreal Protocol at 30: The governance structure, the evolution, and the Kigali Amendment. Comptes Rendus - Geoscience, 350(7), 425–431. https://doi.org/10.1016/j.crte.2018.09.002
Copernicus Atmosphere Monitoring Service. (n.d.). Climate forcing. https://atmosphere.copernicus.eu/climate-forcing
Dupont. (2019). DuPont position statement on Montreal Protocol. https://www.dupont.com/position-statements/montreal-protocol.html
European Environment Agency. (2020). Protecting the ozone layer while also preventing climate change. https://www.eea.europa.eu/themes/climate/ozone-depleting-substances-and-climate-change
Fahey, D. W. (2013). The Montreal Protocol protection of ozone and climate. Theoretical Inquiries in Law, 14(1), 21–42. https://doi.org/10.1515/til-2013-004
Hajost, S. A., & Koehler, J. (1990). The Montreal Protocol: A dynamic agreement for protecting the ozone layer. Ambio, 19(2), 82–86.
McFarland, M. (2009). Perspectives on the roles of science, scientific assessments, the science/policy interface, and industry. In C. Zerefos et al (Eds.) Twenty Years of Ozone Decline (pp. 439-440). Springer.
Molina, M. (1995). Polar ozone depletion: Nobel lecture. https://www.nobelprize.org/prizes/chemistry/1995/molina/lecture/
Molina, M., Zaelke, D., Sarma, K.M., Andersen, S.O., Ramanathan, V., & Kaniaru, D. (2009). Reducing abrupt climate change risk using the Montreal Protocol and other regulatory actions to complement cuts in CO2 emissions. Proceedings of the National Academy of Sciences of the United States of America, 106(49), 20616–20621. https://doi.org/10.1073/pnas.0902568106
Ott, H. (1991). The new Montreal Protocol: A small step for the protection of the ozone layer, a big step for international law and relations. Verfassung Und Recht in Übersee / Law and Politics in Africa, Asia and Latin America, 24(2), 188–208.
Ozone Secretariat. (2020). Kigali Amendment hits milestone 100th ratification, boosting climate action. https://ozone.unep.org/kigali-amendment-hits-milestone-100th-ratification-boosting-climate-action
Salawitch, R.J., Fahey, D.W., Hegglin, M.I., McBride, L.A., Tribbet, W.R., & Doherty, S.J. (2019). Twenty questions and answers about the ozone layer: 2018 Update. Scientific Assessment of Ozone Depletion: 2018. World Meteorological Organization. https://csl.noaa.gov/assessments/ozone/2018/downloads/twentyquestions.pdf
United States Environmental Protection Agency. (n.d.) Basic ozone layer science. https://www.epa.gov/ozone-layer-protection/basic-ozone-layer-science
Weiss, E. B. (2009). The Vienna Convention for the Protection of the Ozone Layer and the Montreal Protocol on Substances that Deplete the Ozone Layer. United Nations.
Additional downloads
You might also be interested in
The Legacies of the Stockholm Conference
Fifty years after Stockholm, we face a triple planetary crisis of climate change, nature and biodiversity loss, and pollution.
The Roots of Forest Loss and Forest Governance
If lessons from past failures on deforestation are learned, forest protection could play a major role in reversing both climate change and biodiversity loss.
India’s State Energy Firms can Boost Energy Security by Progressively Shifting Over INR 2 Trillion Per Year From Fossil Fuels to Clean Energy
New research finds India’s nine state-owned energy companies could progressively redirect a significant share of their over INR 2 trillion annual capital expenditure toward clean and reliable energy, strengthening energy security while accelerating the low-carbon transition.
Joint Letter to Prime Minister Mark Carney on Alberta-Federal MOU
IISD joins five other leading climate and clean energy organizations to highlight the urgent need for a final agreement.