Team October 29, 2018
Step Inside the World’s Freshwater Laboratory’s Laboratory
By Sonya Havens, Research Chemist
The analytical service laboratory
While so much of the work that we do at IISD Experimental Lakes Area takes place right on those famed and picturesque lakes, let’s not forget that all of those samples we collect need to be taken away and analyzed as soon as possible to give us the most accurate picture of what is happening to our lakes.
That all happens in our analytical service laboratory.

QA/QC is a series of procedures used in chemical laboratories around the world, including the analytical service laboratory at IISD Experimental Lakes Area, to ensure they are producing accurate and precise data.
Much of our success and legacy is down to having reliable results and data—all of which stems from a trusted laboratory. In order to achieve those levels to reliability, our analytical service laboratory adheres to the highest standards of quality assurance and quality control.
Always one to open our doors, let us take you on a tour of how we ensure our analysis standards are the highest they can be, and what this means for our freshwater science.
What do we aim for in our analytical service laboratory?
Accuracy. Precision. Consistency.
These three words embody the key objectives of any analytical laboratory.
Accuracy is the measurement of how close an analytical result is to the true value. It is evaluated by comparing the results of an analysis with those of a sample with a known concentration (called a control sample).
Precision refers to how reproducible the results are. We assess precision by measuring a sample multiple times and evaluating how close the results of those measurements are to each other (known as the relative percent difference), regardless of whether or not those measurements are accurate.
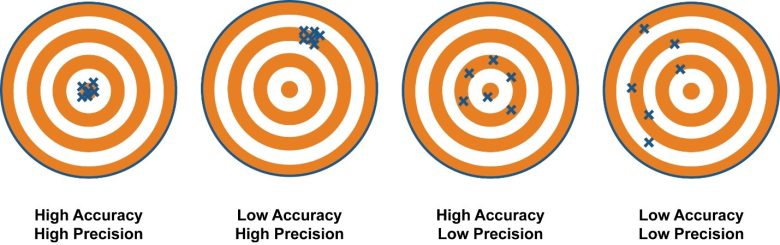
Accuracy. Precision. Consistency. These three words embody the key objectives of any analytical laboratory.
Consistency is the ability to reproduce accurate and precise results throughout time. Our consistency is assessed by including the control sample (this is the sample with a known concentration) in each analysis batch and comparing the results to the known value as well as the values obtained in previous analysis batches.
How do we meet our own high standards?
That’s where Quality Assurance and Quality Control (QA/QC) come in.
QA/QC is a series of procedures used in chemical laboratories around the world, including the analytical service laboratory at IISD Experimental Lakes Area, to ensure that they are producing accurate and precise data.
Quality Assurance is a series of proactive procedures designed to ensure the results of laboratory analyses are accurate and within specified limits of precision. Examples include the incorporation of control samples to assess accuracy and the repeated measurements of the same sample (often referred to as duplicates) to assess precision.
Much of IISD Experimental Lakes Area’s success and legacy is down to having reliable results and data—all of which stems from a trusted laboratory.
Quality Control includes reactive procedures that are designed to identify and correct (if possible) defects in the results of laboratory analyses. It differs from QA in that it entails assessing the data after it has been collected to determine if there are any mistakes, outliers, etc. For example, at the end of each field season, we conduct a trend analysis on each sample site/analysis combination to look for outliers and try to discern the cause of these outliers (e.g., swapped bottles, etc.)
How do we make this happen?
To ensure that the analysis of our samples at the world’s freshwater laboratory are reaching those QA/QC standards, we use a series of methods to check our processes and data.
Here are just a few examples…
Analytical standards are compounds of known concentration and reasonable purity that we used to calibrate an instrument. The samples of unknown concentration are then quantified on the instrument by comparing their response on the instrument to that of the analytical standards.
All this means that we do have to trust that the concentration of the analytical standard is accurate. However, at IISD-ELA we have a rule: trust but verify. To verify that the concentration of the analytical standard is accurate, we purchase standards from two different manufacturers and compare them to each other—yet another step to ensure accuracy.

To ensure that the analysis of our samples at the world’s freshwater laboratory are reaching those QA/QC standards, we use a series of methods to check our processes and data.
We utilize various blanks to evaluate contamination throughout our procedures. These include instrument blanks (ultra pure water run on the instrument), reagent blanks (ultra-pure water with reagents added) and method blanks (ultra pure water that has undergone the entire method). For example, our Total Dissolved Phosphorus analysis is a three-step process:
- Add sulfuric acid and peroxide to a sample and then UV digest.
- Perform a colorimetric reaction by adding chemicals (reagents) that react with phosphorus and result in a colour change that is proportional to the amount of phosphorus in the sample.
- Measure the colour change using a colorimeter or spectrophotometer.
By incorporating a method blank, a reagent blank and instrument blank, we are able to evaluate how much phosphorus is being added to the sample in each of the three steps.
We also incorporate field duplicates, method duplicates and instrument duplicates to assess how much variability is due to each of these stages.
How do we move into the future?
Our analytical service laboratory strives to keep up-to-date with the latest methods and technologies. At the same time, we also need to maintain the integrity of our 50-year dataset.
As we balance advancing into the future with the importance of continuing our ecological legacy, whenever we implement new methods or technologies, we take painstaking steps to ensure that they integrate seamlessly with our current models.
Prior to implementing new methods or technologies, we run samples with both technologies to assess if there will be a “step change” in the data. We then document the quantified “step change” and communicate it to anyone using IISD-ELA data to ensure that the “step change” is not erroneously attributed to something ecological.
For example, we recently received a very generous donation from the Richardson Foundation to purchase a new Auto Analyzer 3 (AA3) to replace our 30-year-old Auto Analyzer 2 (AA2). This new instrument is easier to use, more reliable and can handle more samples, thereby increasing the sample capacity of our laboratory.
As we balance advancing into the future with the importance of continuing our ecological legacy, whenever we implement new methods or technologies, we take painstaking steps to ensure they integrate seamlessly with our current models.
Nevertheless, before this new instrument can be put into action, we need to run the same samples on the old AA2 and on the new AA3 to assess whether they provide the same analytical result. If they do not, we quantify the difference and document it so that it can be communicated to anyone who uses our data.