Research November 29, 2019
Silver Uncontrolled: How nanosilver gets into our fresh water, and what we need to do about it
By Lauren Hayhurst, Fisheries Research Biologist
The Oxford Dictionary describes a ‘silver bullet’ as, “a simple and seemingly magical solution to a complicated problem”, or something that you should have in your arsenal if you want to kill a werewolf.
There are good reasons why silver bullets are notoriously absent from IISD Experimental Lakes Area (IISD-ELA) research and why resulting policy recommendations—the solutions to whole-ecosystem problems such as climate change, eutrophication and oil spills—are complex. It is because they occur on multiple scales and involve a number of factors and responsible parties.
The issue of silver in the environment is another (suitably) silver bullet in freshwater science and policy. We explain its entry into aquatic ecosystems—and what we think needs to happen to protect our ecosystems from it—through the (ab)use of silver puns…
A hundred pieces of (nano)silver
Particles of silver between 1 and 100 nanometres in size are known as “nanosilver” and are widely applied to consumer products and used in medical and agricultural industries. This is because nanosilver is inherently toxic to bacteria and microbes and thus a great antibacterial agent.
Nanosilver is used in agricultural applications from fungal sprays to seed coatings, and in the medical industry from bandages and prosthetics to catheters – products where definitive human health benefits may outweigh potential environmental costs.
Even so, nanosilver is mostly used and abused in an overwhelming number of consumer products. We contribute nanosilver into wastewater every day through wearing and washing underwear, socks, pants, shirts and athletic gear, and using personal care products. You can find it in toothbrushes, shampoos, deodorants, makeup, condoms, yoga mats, dish detergents, frying pans, cutlery, towels, bedding, carpets, vacuum cleaners and washing machines.
This inconspicuous nanosilver is even in ATM buttons, door handles, railings, faucets, cell phones, laptops, keyboards, printer ink AND printer paper! In these instances, it (arguably) serves to entertain society’s favourite past-time of infusing every surface with antibacterial agents, when advocating personal health, hygiene and handwashing would likely take us further as a species.
We don’t mean to nAg
Despite its widespread use, we still don’t fully understand the impact that nanosilver can have on the environment once it enters aquatic ecosystems. While silver itself is regulated in Canada, the increasing and widespread application of nanosilver, and its subsequent release into aquatic ecosystems, is currently unregulated and is largely unknown, unrecognized and unheard of by consumers.

Laboratory studies of the effects of nanosilver on biological organisms and chemical and physical properties have identified multiple ways in which nanosilver is toxic, as it can bind to surfaces and be incorporated into cells.
However, there is little information on the fate and behaviour of nanosilver after it enters the natural environment. Its effectiveness in reducing bacteria and microbes in our clothing, cleaning, exercising and furnishing is decidedly not the effect we desire in our aquatic ecosystems. What about the good bacteria? And the things that eat the good bacteria? And the things that eat those things? Don’t we eat those?

That’s so metal
Researchers wanted to discover what effects nanosilver could have on the health and dynamics of an aquatic ecosystem upon entry, and where else could you intentionally pump kilograms of nanosilver suspended in litres of filtered freshwater into a lake except at IISD-ELA!
Researchers introduced an ‘environmentally-relevant’ amount of nanosilver using a peristaltic pump, hosing and a tote at one site on the shoreline of Lake 222 (to mimic the discharge from a wastewater treatment plant) and added 15 kilograms of nanosilver to a freshwater lake at IISD-ELA over two years, at a rate of 62.5 grams per day.
They found that a majority of nanosilver settled to the sediments, with approximately half the total silver load present in the sediments by the end of each of the two nanosilver addition years. Despite this sinking and settling, nanosilver was not tightly bound to the sediments and was re-introduced into the water column after the ice melted in the spring each year. Nanosilver effectively spread horizontally and vertically throughout the lake from the shoreline site from which it had been added, where it persisted for the duration of the study, and interacted with biological organisms as well as chemical and physical properties of the lake.
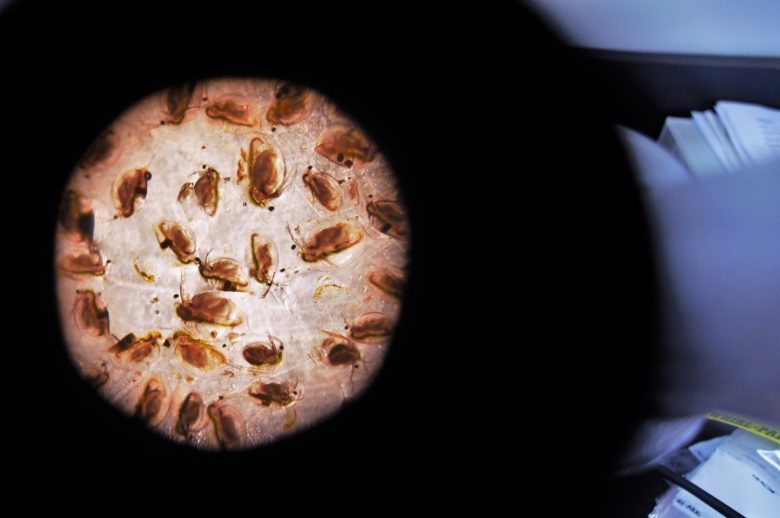
Results of these nanosilver additions were mixed across levels of the food chain; while algae, bacteria, benthic invertebrates and zooplankton showed few short-term impacts and were largely unaffected by the nanosilver that we added to the lake, significant long-term results were observed in fish across cellular, individual and population levels.
We looked for the “silver lining”…and found it on fish gills and guts
We found that silver bioaccumulated in fish tissues through both respiratory and dietary sources, and that perch suffered from oxidative stress at the cellular level. Energetic models of perch revealed declines in their prey consumption and lower metabolic rates.
There were declines in perch populations over the course of the study, even though their main pike predators shifted their feeding behaviours from nearshore to offshore locations and experienced stunted growth (stay tuned!). Even four years after they stopped adding nanosilver to the lake, researchers still found significant concentrations of silver in the tissues of the pike that had been exposed to nanosilver.

Hi-ho, Silver! Away!
What we found has implications for the use of nanosilver in consumer products and the health of freshwater ecosystems across the globe.
We have evidence of short-term impacts on fish after only two months of adding nanosilver and long-term impacts two years after environmentally-relevant nanosilver additions. We also found that those impacts continued to manifest even four years after we stopped adding nanosilver to the lake.
It’s important to note that we stopped adding nanosilver to the lake after two years, which is not the case in the natural environment where nanosilver continues to enter aquatic ecosystems at greater-than-ever quantities.
“Only YOU can prevent nanosilver release” – Smokey the Fish
The Government of Canada needs to revise their Canadian Water Quality Guidelines for the Protection of Aquatic Life to regulate nanosilver as it regulates silver, with continued and improved monitoring of nanosilver in aquatic ecosystems. Additionally, the introduction of mandatory labelling of nanoproducts would help consumers make informed choices.

While we wait for that to happen, the vast and growing extent of nanosilver in clothing and materials can be addressed at the consumer level by embracing to the eco/sustainable/slow fashion movement. By reducing the quantity of new clothing (capped with nanosilver) that we purchase, researching sustainable and ethical brands, and recycling clothing, we can make a conscious change that will benefit not only the fish in a lake but also improve social and environmental standards on a global scale.
Researchers in this whole-ecosystem nanosilver study included graduate students and principal investigators from Trent University, Lakehead University, and Institut national de la recherche scientifique. Research internships were provided through support from an NSERC CREATE program (CREATE H2O), coordinated by the University of Manitoba. Funding for the whole-lake addition project was provided by the Natural Sciences and Engineering Research Council of Canada through the Strategic Grants Program, and matching grants from Environment and Climate Change Canada. IISD-ELA provided in-kind support for the project, Support for biomarker analyses was provided by the National Contaminants Advisory Group of Fisheries and Oceans Canada. Mercury, energy density analyses, and bioenergetics modelling was supported by infrastructure funded by the Canadian Foundation for Innovation, NSERC Discovery, and support from IISD-ELA and the Mitacs Accelerate Program.
You know that ground-breaking freshwater research you just read about? Well, that’s actually down to you.
It’s only thanks to our generous donors that the world’s freshwater laboratory—an independent not-for-profit—can continue to do what we do. And that means everything from explore what happens when cannabis flushes and oil spills into a lake, to how we can reduce mercury in fish and algal blooms in fresh water—all to keep our water clean around the world for generations to come.
We know that these are difficult times, but the knowledge to act on scientific evidence has never been more important. Neither has your support.
If you believe in whole ecosystem science and using it to bring about real change to fresh water around the globe, please support us in any way you are able to.